Agarose gel electrophoresis is an electrophoresis method that uses agarose as a support medium. It is mainly used for DNA cutting gel recovery, DNA separation, and proving whether the DNA is recombined and whether the plasmid is cut successfully. So waht is agarose gel whattrophoresis? and how does it work? What are the keys to successful agarose gel electrophoresis?
1. What is agarose gel electrophoresis
2. How does it work?
3. What are the factors affecting agarose gel electrophoresis?
3.1 Agarose
3.2 Nucleic acid dyes
3.3 DNA marker
3.4 PH value of electrophoresis buffer
3.5 Product performance test
4. Related product information and client feedback
5. Published references (using nucleic acid electrophoresis products of Yeasen)
1. What is agarose gel electrophoresis
Electrophoresis means that the charged test sample swims in the direction of the corresponding electrode under the action line of the electric field in the inert support medium at different speeds, and the components will be separated into narrow zones. Appropriate detection methods It can record the electrophoretic zone map, or calculate the percentage content. Electrophoresis mainly includes free electrophoresis and zone electrophoresis. The main difference is that the former has no support and the latter has support. Agarose gel electrophoresis belongs to the gel support zone electrophoresis in zone electrophoresis. Agarose is the part of agar that forms a three-dimensional gel matrix composed of helical agarose molecules. These agarose molecules are held in place by hydrogen bonds in the supercoil bundle, with channels and pores through which the molecules pass. The percentage of agarose in the gel affects the size of the pores, which in turn affects the size and velocity of the molecules that can pass through. The higher the agarose percentage, the smaller the pore size, and the smaller and slower the molecules that can pass through.
Agarose gel electrophoresis is simple and fast, and samples can be electrophoresed without prior treatment. In addition, the structure of agarose is uniform, and it is approximately free electrophoresis, so the sample diffusion is relatively free. Agarose gel electrophoresis has very little adsorption to the sample, so the electrophoresis pattern is clear, with high resolution and good repeatability. Agarose gel is transparent, without ultraviolet absorption, and the electrophoresis process and results can be directly detected and quantitatively determined by ultraviolet light. After electrophoresis, the zone is easy to stain, the sample is easily eluted, and it is convenient for quantitative determination. However, its mechanical strength is poor, it is easy to break, and the concentration cannot be too low. Therefore, agarose which is not easy to break can be used as a basis to judge the quality of agarose. In addition, it is easy to be contaminated by bacteria and inconvenient to store. The separation is not as good as PAGE electrophoresis, which can distinguish DNA fragments that differ by 1bp.
2. How does it work?
The main difference between the analysis principle of agarose gel electrophoresis and other support electrophoresis is that it has the dual functions of "molecular sieve" and "electrophoresis". The agarose gel, as a molecular sieve, has network structure, and the material molecules will be resisted when passing through. The DNA molecules of different sizes are charged differently, and the force in the electric field is also different, so the speed of migration from the negative electrode to the positive electrode is unequal. When running, the macromolecular substances are great resistance. So in gel electrophoresis, the separation of charged particles depends not only on the nature and quantity of the net charge, but also on the size of the molecule, which greatly improves the electrophoretic resolution.
3. What are the factors affecting agarose gel electrophoresis?
These three items are necessary: Agarose, Nucleic Acid Dye & DNA Marker.
3.1 Agarose
Agarose is a purified linear galactan hydrocolloid, extracted from agar or agar-containing algae, and is structurally a linear polymer linked by β-D-galactopyranosine (1-4) and 3,6-Anhydro α-L-galactopyranosyl. The basic property of agarose, include:
- Sulfate content - purity index;
- Gel strength - the external force applied to the gel to break it;
- Gel point - the temperature at which a water-soluble agarose solution forms a gel after cooling;
- Electroendosmosis (EEO) - an electrokinetic movement of a liquid through a gel. The anionic groups in the agarose gel adsorb on the matrix without migration, but the dissociated cations migrate toward the negative electrode, resulting in electroosmosis. Since the electrophoretic migration of the sample is usually towards the positive electrode, the internal convection generated by the EEO can interfere with the separation efficiency.
Therefore, according to the basic properties of agarose, a high-quality agarose gel should have clear wells, with high purity (low sulfate content), high gel strength (not be easily broken), relatively high gel point (fast solidification at room temperature), and low electro-endosmosis, etc. The optimum agarose gel concentration for different sizes of target fragments is not same. The distance DNA fragment migrates (mobility rate) is inversely proportional to the number of base pairs. The longer DNA fragment is, the greater resistance meet. And the efficiency of the macromolecules passing through the pore size of the gel is lower than that of the smaller molecules, so the migration of large molecules is slow. Generally speaking, the larger charge of the molecule, the smaller diameter, and the faster the electrophoretic migration of the molecule whose shape is closer to a spherical shape. And the greater the gel concentration, the smaller the molecular pore size, the lower the DNA mobility, the higher the resolution, and the smaller DNA fragments can be separated. Therefore, choosing an appropriate gel concentration is conducive to the successful operation of agarose gel electrophoresis. So how to choose the right gel concentration? According to the principle that high agarose gel concentration is suitable for small fragments, you can refer to the following table to find the best gel concentration for you.
Table 1. Agarose Gel Concentration
| Agarose Gel Concentration % | ≥3 | 2-3 | 1-2 | 0.7-1 | ≤0.7 |
| Fragments bp | ≤200 | 200-700 | 700-1500 | 1500-5000 | ≥5000 |


Figure 2. Agarose 10208ES60 from Yeasen
3.2 Nucleic acid dyes
Agarose gel electrophoresis is inseparable from the support of nucleic acid dyes. Ethidium bromide (EB) is the earliest and most mature nucleic acid dye, and it was widely used by experimenters in the early days because of its low price and high sensitivity. However, its molecular weight is small, and it can easily penetrate the cell membrane, which is a mutagen and with high carcinogenicity. The potential harm to the human body is very great, so experimenters need to be cautious enough when using it. In addition, the disposal of waste glue and waste liquid is time-consuming, laborious and expensive, and the things that accidentally touch the EB area may no longer be used, which is a love-hationship.
With the development of technology and people's attention to health and environmental safety, non-toxic nucleic acid dyes came into being.
So, is the non-toxic nucleic acid dye claimed on the market non-toxides non-toxic, and what are the advantages? With these problems, you may refer to YeaRed (Cat#10202), a safe and non-toxic nucleic acid dye that replaces EB, as a basis for selection.
YeaRed is a new type of non-toxic nucleic acid dye with a unique oily macromolecular structure developed by Yeasen Biotechnology (Shanghai) Co.,Ltd., which is not able to penetrate the cell membrane and enter the cell, and does not easy to volatilize and sublime, and the human body will not inhale it, thus ensuring the safety of the experimenter to the greatest extent. In addition, YeaRed nucleic acid dye was tested by Mini-Ames, and the result was negative. That is, YeaRed was non-toxic under the gel staining concentration. (For the test report, please contact our company for technical consultation)

Figure 3. YeaRed (Cat#10202) from Yeasen
3.3 DNA marker
DNA Marker is a combination of DNA fragments with different molecular weights. It is mainly used for the electrophoresis of DNA when they are added to the gel together with the sample for electrophoresis separation. It can be roughly estimated the molecular weight of the sample DNA and its concentration by referring to the size and brightness of the sample fragments of the DNA Marker. What are the features of a DNA Marker that can complete agarose gel electrophoresis? As follows:
- Strong stability, can be stored at room temperature for 3-6 months;
- The background is clean, the shape is stable, and the size is accurate;
- The concentration of each fragment is known and with indicator fragment, which is convenient for positioning and semi-quantitative analysis;
- The loading buffer is already included in the DNA marker and can be directly electrophoresed, which is convenient and quick;
- 5× loading buffer is attached, which can be used for sample loading.
At present, the DNA Marker of Yeasen Biotechnology (Shanghai) Co.,Ltd., covers DN A fragments from 50 bp to 15 kb, which meets the needs of most experiments.

Figure 4. Product feat features
3.4 PH value of electrophoresis buffer
In addition to the three points mentioned above, the pH value of the buffer is also one of the key factors affecting the success of agarose gel electrophoresis. The ionic strength of the solution is too low, the conductivity will be reduced, and the DNA will be completely immobile or migrate very slowly. When the ions are too strong, the conductivity increases, which will generate a lot of heat energy, which will change or even melt the gel, thereby denaturing the DNA.
3.5 Product performance test
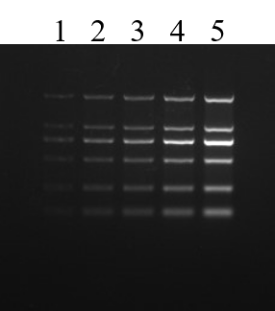
Figure 5. Agarose gel electrophoresis of the nucleic acid dye YeaRed. Electrophoresis conditions: 120 V, 30 min. The sample is DNA Marker (DL2000), and the loading volume of wells 1-5 is 1-5 uL, respectively.
4. Related product information and client feedback
4.1 Related product information
The products provided by Yeasen are as follows.
| Product Usage | Product Name | Cat# | Size |
| Agarose Gel Electrophoresis | Agarose | 10208ES60 | 100 g |
| DNA Marker | GoldBand DL2000 DNA Marker | 10501ES60 | 100 T |
| GoldBand DL5000 DNA Marker | 10504ES60 | 100 T | |
| GoldBand DL10,000 DNA Marker (Inquire) | 10505ES60 | 100 T | |
| GoldBand 100bp DNA Ladder | 10507ES60 | 100 T | |
| GoldBand 1 kb DNA Ladder | 10510ES60 | 100 T | |
| GoldBand 50 bp DNA Ladder (Inquire) | 10515ES60 | 100 T | |
| GoldBand 100 bp plus DNA Ladder (Inquire) | 10516ES60 | 100 T | |
| GoldBand 200 bp DNA Ladder (Inquire) | 10517ES60 | 100 T | |
| GoldBand 500 bp DNA Ladder (Inquire) | 10518ES60 | 100 T | |
| Nucleic Acid Dyes | YeaRed™ Nucleic Acid Gel Stain (10,000× in Water) | 10202ES76 | 500μl |
4.2 Client feedback
- From Fudan University
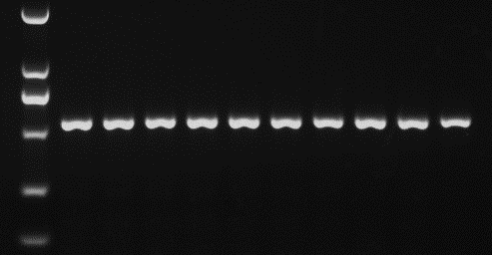
Figure 6. Agarose gel electrophoresis of the nucleic acid dye YeaRed. Electrophoresis conditions: 120 V, 30 min. Marker: DL2000. Target fragment size: 550 bp.
- From Instithe tute of Hematology, Chinese Academy of Medical Sciences
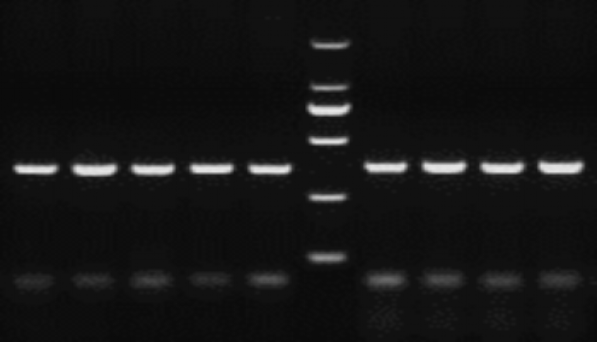
Figure 7. Agarose gel electrophoresis of the nucleic acid dye YeaRed. Electrophoresis conditions: 150 V, 30 min in 2% agarose gel. Marker: DL2000. Target fragment size: 350 bp.
5. Published references (using nucleic acid electrophoresis products of Yeasen)
[1]Wang Y, Fu Z, Li X, et al. Cytoplasmic DNA sensing by KU complex in aged CD4+ T cell potentiates T cell activation and aging-related autoimmune inflammation. Immunity. 2021;54(4):632-647.e9. doi:10.1016/j.immuni.2021.02.003 (IF:31.745)
[2]Yang X, Gao F, Zhang W, et al. "Star" miR-34a and CXCR4 antagonist based nanoplex for binary cooperative migration treatment against metastatic breast cancer. J Control Release. 2020;326:615-627. doi:10.1016/j.jconrel.2020.07.029 (IF:7.727)
[3]Qiao Y, Du J, Ge R, et al. A Sample and Detection Microneedle Patch for Psoriasis MicroRNA Biomarker Analysis in Interstitial Fluid. Anal Chem. 2022;94(14):5538-5545. doi:10.1021/acs.analchem.1c04401 (IF:6.986)
[4]Lin Q, Ye X, Huang Z, et al. Graphene Oxide-Based Suppression of Nonspecificity in Loop-Mediated Isothermal Amplification Enabling the Sensitive Detection of Cyclooxygenase-2 mRNA in Colorectal Cancer. Anal Chem. 2019;91(24):15694-15702. doi:10.1021/acs.analchem.9b03861 (IF:6.350)
[5]Lin Q, Huang Z, Ye X, et al. Lab in a tube: Isolation, extraction, and isothermal amplification detection of exosomal long noncoding RNA of gastric cancer. Talanta. 2021;225:122090. doi:10.1016/j.talanta.2021.122090 (IF:6.057)
[6]Liu C, Zou G, et al. 5-Formyluracil as a Multifunctional Building Block in Biosensor Designs[J]. Angew Chem Int Ed Engl. 2018 Jul 26;57(31):9689-9693. (IF 11.992)
[7]Wang M, Zhang S, Zheng G, et al. Gain-of-Function Mutation of Card14 Leads to Spontaneous Psoriasis-like Skin Inflammation through Enhanced Keratinocyte Response to IL-17A. Immunity. 2018;49(1):66-79.e5. doi:10.1016/j.immuni.2018.05.012 (IF:19.734)
[8]Zhang Y, Ding H, Wang X, et al. MK2 promotes Tfcp2l1 degradation via β-TrCP ubiquitin ligase to regulate mouse embryonic stem cell self-renewal. Cell Rep. 2021;37(5):109949. doi:10.1016/j.celrep.2021.109949 (IF:9.423)
[9]Lin Q, Ye X, Yang B, et al. Real-time fluorescence loop-mediated isothermal amplification assay for rapid and sensitive detection of Streptococcus gallolyticus subsp. gallolyticus associated with colorectal cancer[J]. Analytical and bioanalytical chemistry, 2019, 411(26): 6877-6887. (IF6.35)
[10]Lin Q, Ye X, Huang Z, et al. Graphene Oxide-Based Suppression of Nonspecificity in Loop-Mediated Isothermal Amplification Enabling Sensitive Detection of Cyclooxygenase-2 mRNA in Colorectal Cancer[J]. Analytical chemistry, 2019. (IF6.35)
[11]Wang W, Nie A, Lu Z, et al. Catalytic hairpin assembly-assisted lateral flow assay for visual determination of microRNA-21 using gold nanoparticles[J]. Microchimica Acta, 2019, 186(9): 661. (IF5.479)
[12]Cheng H, Fan X, et al. Cyclodextrin-Based Star-Like Amphiphilic Cationic Polymer as a Potential Pharmaceutical Carrier in Macrophages[J]. Macromol Rapid Commun. 2018 May 28:e1800207. (IF 4.441)